Abstract
Crystallization is a critical operation in the fertilizer industry, governing the physical and chemical properties of fertilizers. It ensures controlled nutrient release, mechanical strength, solubility, and storage stability, while enabling efficient production of MAP, DAP, MKP, AS, and NPK fertilizers.
Introduction
India remains one of the largest importers of fertilizers globally, with substantial volumes brought in to meet agricultural demand. In FY 24-25, Fertilizer imports was around 160 Lakhs MT valuing USD 7.71 billion. The major import was of Mono and Di-ammonium phosphate (MAP, DAP), MOP (Muriate of Potash), NPK, Urea, etc. There is also growing demand for water soluble (WSF) and Super speciality grade fertilizers (SSF) in cash crop and horticultural crops. Total import in FY 23-24 was 3.35 Lakh MT from China, Belgium, Egypt, etc. Basically, all these WSF and SSF fertilizers are nothing but pure form of NPK fertilizers which is obtained by blending technical grade MAP, MKP, Calcium or ammonium nitrate, ammonium sulphate, Potassium sulphate, and urea. All these fertilizers are of solid and crystalline in nature wherein the physical properties such as particles size and structure are governed by crystallization process during manufacturing process.
Crystallization is widely used unit operation in fertilizer industry. It has many applications starting from manufacturing of primary raw material, intermediate products to finished products. Physical properties of there materials such as moisture content, particles size, hygroscopicity, stacking and crushing strength, etc plays vital roles in processing, handling, transportation, bagging, storage and application. Crystalline structure affects the solubility of nutrients. Fertilizers with well-formed crystals can offer slow or controlled release, improving nutrient uptake by plants and reducing leaching losses. Crystallization enhances the mechanical strength of granules. Stronger granules reduce dust formation, improve handling, and ensure uniform application in the field. Proper crystallization helps to achieve consistent granule size, which is essential for even spreading during application and predictable dissolution rate. Crystalline fertilizers are generally less hygroscopic than amorphous ones. This reduces caking and moisture absorption, improving shelf life and storage conditions. Crystallization is often part of cooling or evaporation steps in fertilizer production. Efficient crystallization can reduce energy consumption, improve yield, and minimize waste. By controlling crystallization conditions (temperature, concentration, additives), manufacturers can tailor fertilizers for specific crops, soils, or climates. There is diverse range of granular as well as water soluble fertilizers that are produced from crystallization such as Nitrate based fertilizers, Ammonium sulphate, Mono and dia- ammonium phosphate, Potash, Potassium sulphate, etc.
Crystallization
Crystallization is based on the transformation of a substance from a liquid or dissolved state into a solid crystalline form. Crystallization occurs when a solution becomes supersaturated, meaning it contains more dissolved material than it can hold at a given temperature. This leads to the formation of crystals as the excess solute precipitates out. It’s a four-step process, namely, supersaturation, nucleation, crystal growth, separation and drying. All these steps are governed by temperature, solvent choice, rate of cooling and presence of impurities. Crystallization is critically important in the fertilizer industry for both technical and economic reasons. Crystallization helps remove impurities from raw materials like phosphoric acid, ammonium salts, and potassium compounds. This also ensures high nutrient content and consistent chemical composition in fertilizers like MAP, MKP, and AS. The particles size of the crystals controls the nutrient release rate. Proper crystallization improves mechanical strength of the crystals or granules. It avoids dusting, caking and particles crushing during material handling. In water soluble fertilizers, precise crystallise is require ensuring complete fertigation and avoid pipe clogging. Typical crystallization properties are shown in Table 1.
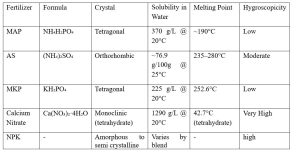
Table 1: Crystallization Properties of few Fertilizers
Crystallizer Overview in Fertilizer Sector
In fertilizer industry, crystallizers are essential for recovering solid nutrient from liquid solution, purifying fertilizers, improving fertilizer quality and reducing waste. Following are few examples of types of crystallizers used in fertilizer industry (Table 2).
Reactive Crystallization
Reactive crystallization is a process where crystallization occurs simultaneously with a chemical reaction, rather than as a separate downstream step. Product precipitates as crystals during the chemical reaction due to supersaturation created by the reaction itself. Combining reaction and crystallization in one step, reduces equipment and energy costs. However, it requires precise control to avoid fines, agglomeration or poor crystal quality. This type of crystallizer is particularly useful in fertilizer manufacturing when the product forms directly as a solid from the reaction mixture such as MAP and DAP production.
NH4H2PO4, mono-ammonium phosphate (MAP) and (NH4)2HPO4, di-ammonium phosphate (DAP)have become leading phosphate fertiliser products worldwide. Ammonium phosphates are produced by reactions of ammonia and phosphoric acid resulting in the formation of the mono-basic, di-basic, or tri-basic salts. Inefficiencies in the MAP/DAP production process are due to the lack of a fundamental understanding of the crystallisation–reaction mechanisms. The reaction is exothermic, so the MAP and DAP are produced at an elevated and relatively constant temperature of 110 °C. The mono-ammonium phosphate (MAP) tends to produce needle forms of crystals while the di-ammonium phosphate (DAP) results in more granular forms of crystals. Tri-ammonium phosphate (TAP—(NH4)3PO4) is an unstable crystal and less soluble than MAP and DAP. Ammonium phosphate production process which needed a high quality Phos acid in the process. A high impurity content that can cause a scaling and blocking problem in the acid concentrator heater. The impact of impurities on the ammonium phosphates slurry characteristics causes several problems in mixing, flow ability and pumping efficiency. These factors contribute to the unit process/plant efficiency. Hydrolysis of metal ions (Fe3+ and Al3+) initiate solids formation as they act as nucleation sites for crystallisation. In industry, the hydrolysis process leads to some challenges and causes inefficiencies. This is due to a high recycle ratio in the granulation process, the fluidity of the slurry, and uncontrolled product size distribution, which further triggers a domino effect that can decrease the downstream unit’s efficiency and produce an off specification product. Reactive crystallizers are now commonly used in MAP and DAP production processes.
Cooling Crystallizer
Cooling crystallizers are essential devices used in various industries to promote the controlled formation of crystals from supersaturated solutions by gradually lowering temperature. A cooling crystallizer is a type of crystallizer where temperature reduction is used to induce supersaturation, leading to the formation of crystals. Solubility of the solute decreases as temperature drops, causing crystals to form. Cooling is usually provided via Indirect cooling via jackets, coils, or heat exchangers, sometimes direct cooling with chilled water. Controlling parameters such as temperature gradient, residence time, agitation, and feed concentration needs to be maintained during operation. Crystal growth is mainly controlled by cooling rate and mixing intensity. It’s commonly used in the production of fertilizers like Mono Potassium Phosphate (MKP), where solubility decreases significantly with temperature.
Potassium hydroxide (KOH) reacts with phosphoric acid (H₃PO₄) to form MKP. MKP has high solubility and its solubility is strongly temperature-dependent. The solution is cooled to around 10°C, where MKP begins to crystallize. Cooling the solution leads to supersaturation, which triggers crystal formation. This method allows for controlled crystal size, high purity, and efficient recovery. Crystals are separated from the mother liquor via filtration or centrifugation. Crystals are dried to produce the final product. The controlled environment of cooling crystallizers allows for the production of high-purity crystals, which is vital in many applications. The crystallization process consists of two major steps, nucleation and crystal growth which continue to occur simultaneously, and their rate is driven by the existing supersaturation in the solution. Supersaturation can be altered by changing the operating conditions. Depending upon the conditions, either nucleation or growth may be predominant over the other, and as a result, crystals with different sizes and shapes are obtained. As the time progresses, supersaturation level decreases and a stage of equilibrium is reached where the crystallization process reaches a steady state. The cooling rate significantly impacts crystal size, shape, and purity. Rapid cooling generally produces many small crystals, while slow cooling delays the operation leading into high operational cost. There are various types of equipment used in Industrial practice such as jacketed stirred tanks, trough crystallizers, Oslo crystallizers, etc.
Fluidized Bed Crystallizer
A fluidized bed crystallizer, also called as Oslo Crystallizer, is a specialized type of crystallizer designed to produce uniform, coarse crystals by maintaining a fluidized state of growing crystals within the crystallization zone. Crystallization begins when the solution becomes supersaturated, meaning it contains more dissolved solute than it can hold at equilibrium. Supersaturation is typically achieved by cooling, evaporation, or chemical reaction. Crystals are kept in suspension by an upward flow of liquid (mother liquor). This creates a fluidized bed, where crystals are free to move and grow without settling. Seed crystals in the bed grow as solute molecules deposit on their surfaces. The uniform flow and mixing prevent agglomeration and promote even crystal growth. It is particularly useful in fertilizer industries for crystallizing salts like potassium chloride, ammonium sulphate, and in some advanced NPK processes.
As a fertilizer, AS supplies two fundamental nutrients: nitrogen and sulphur. Of the total amount of nitrogen fertilizers only 4% is ammonium sulphate. Recently, the world-wide supply of AS has increased due to the production by direct reaction crystallization from (spent) sulfuric acid and ammonia. In process initially crystallizer is fed with saturated solution of ammonium sulphate. Water is being evaporated to increase supersaturation. Seed crystals of ammonium sulphate is introduced in the system to initiate controlled growth of crystals. Solvent evaporation continues till solution reaches the supersaturation level. Maintaining the supersaturation condition of the system is the critical parameter in avoiding the uncontrolled growth of crystals. Residence time is optimised for desired crystal size. Once crystallization completed, the crystals are being recovered from recovery unit. Once system reaches steady state, feed saturated with Ammonium sulphate solution is continuously fed to the system and crystals are recovered continuously through the recovery unit.
Granulators
Dependant on the desired growth patterns, a wide variety of fertilizers are produced, ranging from single nutrient to multi-nutrient fertilizers. Single nutrient fertilizers that only produce N include ammonium nitrate, calcium ammonium nitrate (CAN) and urea. At the same time, triple superphosphate (TSP) and single superphosphate (SSP) only provide P. Meanwhile, fertilizers such as mono-ammonium phosphate (MAP), di-ammonium phosphate (DAP) and urea superphosphate (USP) provide both N and P. Finally, NPK fertilizers are those that provide crops with all three nutrients: N, P and K. There are two main categories of NPKs, depending on how their constituents are combined. They can be produced, for example, by physically mixing together separate granules of ammonium nitrate, MAP and muriate of potash (MOP) in a process known as bulk blending. On the other hand, compound fertilizers can also be produced using a granulation process to combine all three nutrients homogeneously within a single granule. Compound NPKs are generally preferred over NPK blends as they do not segregate during storage and transportation, helping to ensure that nutrients are spread evenly during field application. Furthermore, compound NPKs can be manufactured using a range of different raw materials. The N source is usually a mix of ammonia, ammonium nitrate or urea. However, ammonium sulfate can also be used, either sourced as an external product or produced in-situ from sulfuric acid and ammonia. The P can be sourced from phosphoric acid. Most of the NPK production process are based on pipe reactors, a production technology that offers distinct advantages in terms of flexibility, safety and cost. A granulation plant using a single pipe reactor process comprises a drum granulator, a drum dryer, screens and crushers, cooling equipment and, if required, a coating drum. The different solid raw materials are fed directly to the granulator along with the solids recycled from the previous process. Afterwards, the liquid raw materials are fed mainly to the pipe reactor, where the phosphoric acid is neutralised by the ammonia. The molar ratio between the phosphoric acid and ammonia fed to the pipe reactor is an important parameter that needs to be considered when tuning the pipe reactor. Some ammonia is also fed into the granulator throughout an ammonia sparger, in order to help adjust the composition of the fertilizer and to assist the granulation process. The granulated product is further dried in drum dryer with co-current flow of hot air. A lump screen at the outlet of the dryer separates large agglomerates and directs these to the lump breaker. This device protects the elevator located at the outlet of the dryer from the risk of blockage and damage. NPK Granules are then passed over the screen. The granules of uniform size follows the process and undersized and oversized particles are recycle back to the process.
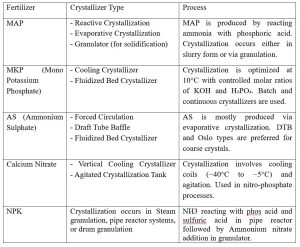
Table 2: Crystallizers used in Fertilizer Industries
Conclusion
Crystallization plays a vital role in producing high-quality fertilizers with controlled nutrient release, consistent granule strength, and improved storage stability. By optimizing crystallizer types and operating conditions, fertilizer manufacturers can enhance efficiency, product purity, and overall process reliability.































