Abstract
Green chemistry processes aim to utilize renewable sources using benign catalysts to manufacture chemicals and fuels. Catalysts are crucial for producing highly selective products by enhancing reaction efficiency and reducing energy consumption. Designing catalysts that do not involve toxic chemicals is essential to align with the principles of green chemistry. The article provides valuable insights into the heterogeneous catalysts employed in various chemical processes, contributing to the achievement of sustainable goals.
Introduction
The United Nations’ sustainable development goals and the twelve principles of green chemistry emphasize responsible production processes vital for future economic circularity. Industries are now choosing these cost-effective processes that generate less waste, ultimately reducing disposal and treatment needs. In this context, using abundant renewable biomass as a feedstock can support sustainable development goals by replacing non-renewable energy sources that raise environmental concerns. Green chemistry identified the application of catalysis for various biomass conversion processes as one of the principles used to enhance reaction efficiency with low energy requirements. Catalysts can make processes more economically viable by allowing lower reaction conditions, such as reduced temperature and pressure, which can decrease costs. Consequently, the energy sectors utilize catalytic conversion processes in petroleum refineries, produce hydrogen from biomass, and employ carbon capture, utilization, and sequestration.
Catalysts are primarily classified into two categories: homogeneous catalysts and heterogeneous catalysts. Homogeneous catalysts, known for their firm activity and high selectivity, are limited in practical use due to the difficulty of separating them from the reaction mixture. Therefore, the researchers are more interested in environmentally benign heterogeneous catalysts due to their ease of separation and recycling. Catalysts can be tailored with the required active sites through their preparation methods to enhance specific reaction activity. The article offers an overview of important chemical processes that use heterogeneous catalysts. The role of catalysts in facilitating green chemical processes is illustrated in Fig. 1. Among the various catalytic processes, the production of sustainable aviation fuels (SAF) through biomass valorization, eco-friendly methods for synthesizing ketones via ketonization, and the utilization of CO2 in the synthesis of value-added products.
Biomass Valorization: Production of Sustainable Aviation Fuel (SAF)
The synthesis of SAF from biomass derivatives has attracted attention due to its high selectivity achieved under moderate reaction conditions. Currently, the researchers focus on transforming platform chemicals derived from lignocellulose biomass, an abundantly available feedstock that is cost-effective, easily accessible, and does not compete with food sources. However, SAF production is more challenging due to the particular hydrocarbon composition required, the complexities associated with multiple reaction steps, and issues related to catalyst stability. The selective conversion of biomass-derived compounds, particularly furanic products, into fuels and fuel additives has significant commercial potential with environmentally friendly characteristics.
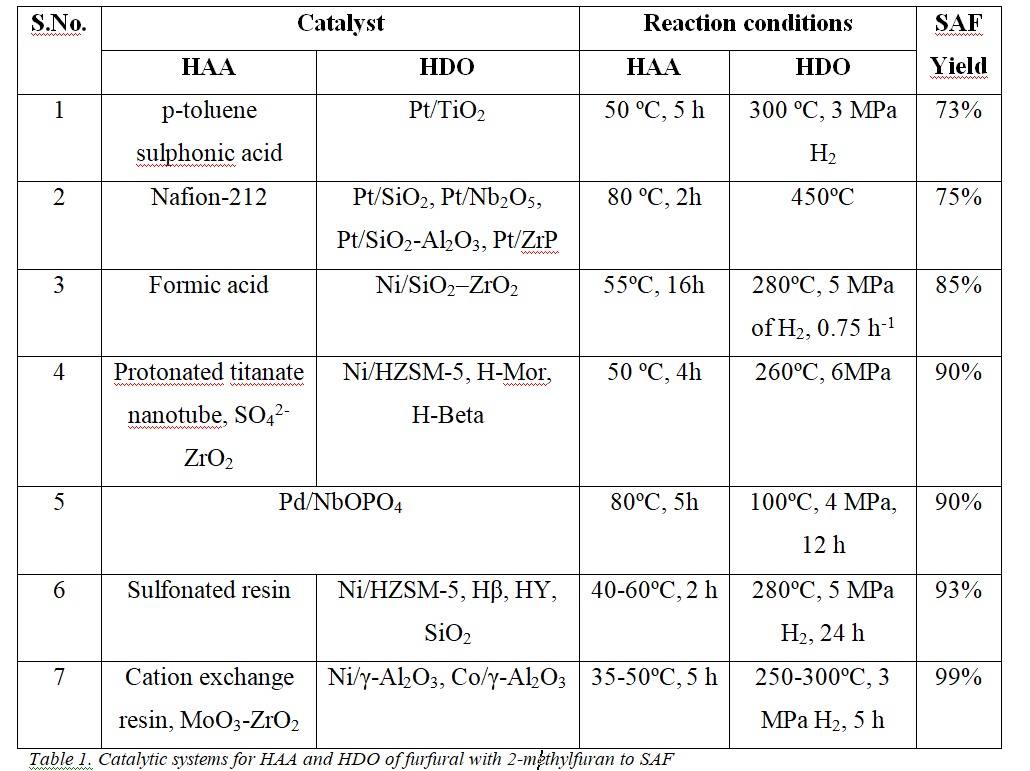
The SAF production process includes two-step hydroxy alkylation-alkylation (HAA) and hydrodeoxygenation (HDO) reactions, as displayed in Fig. 2. HAA appears between an aldehyde furfural, andα-hydrogen compound 2-Methylfuran in the presence of an acid catalyst to produce a C15biofuel precursor. HAA on various solid acid catalysts like heteropolyacids, resins (Amberlyst-15, Amberlyst-36, Nafion-212, and Nafion-115), zeolites (HZSM-5, HY, and H-USY), and acid functionalized materials were reported by several research groups[1]. Nafion-212 resin exhibited the utmost catalytic activity of 75% conversion of 2-Methylfuran among the catalysts[2]. Consequently, the HAA reaction depends on the acidity of the catalyst, making high acidity essential for achieving great purity in the biofuel precursor. The large pore size of mesoporous catalysts aids in diffusing bulky reactant and product molecules, enhancing accessibility and improving activity[3]. Later, the MoO3-promoted mesoporous ZrO2 catalyst has recently demonstrated high catalytic activity, achieving ~85% conversion of 2-Methylfuran at low reaction temperatures of 50 ºC. This superior catalytic activity was attributed to the catalyst’s firm acidity of tetragonal phases[4].
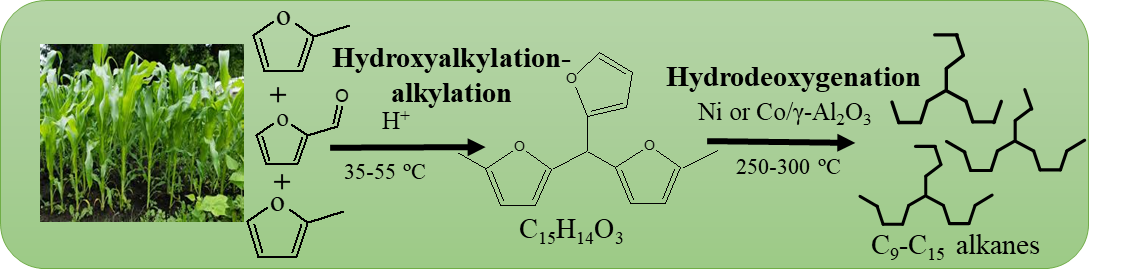
The HDO is a second-step process essential for removing oxygen in the presence of H2 pressures from the C15 precursor, resulting in a high selectivity of jet-fuel range hydrocarbons (C9-C15), also called SAF. Previously, the HDO study was extensively conducted on noble metal catalysts such as Pt, Pd, and Ru supported on C, C+H-Beta, and γ-Al2O3[5]. The alkanes obtained over these noble metal catalysts exhibited excellent fuel properties of 0.7896 g/ml density and -100 ºC pour point[2]. Cost-effective transition metals such as Ni and Co were introduced to reduce reliance on these expensive noble metals[4]. These non-noble metal-supported catalysts achieved complete oxygenated compound conversion to C9-C15hydrocarbons, with C14being the major hydrocarbon. Table 1 presents an overview of different catalysts employed to synthesize SAF.
Ketonization: A Green Catalytic Process for the Synthesis of Ketones
In general, ketones are prepared by well-known Friedel-Crafts acylation reactions using acid chlorides as an acylating agent over an AlCl3 catalyst. Ketones are versatile compounds that can be converted into fuels and chemicals. Using corrosive acid chlorides, acid anhydrides, and catalysts like AlCl3 is hazardous and creates a lot of environmental issues. In contrast, ketonization is a green pathway toward converting carboxylic acids to ketones with CO2 and water as byproducts over solid metal oxide catalysts, as shown in reaction scheme 1[6].
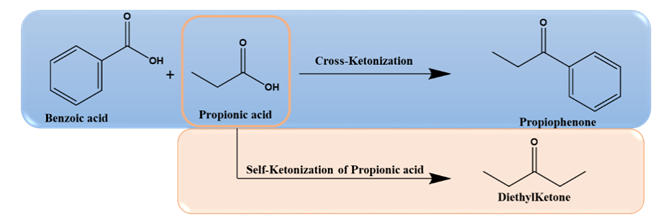
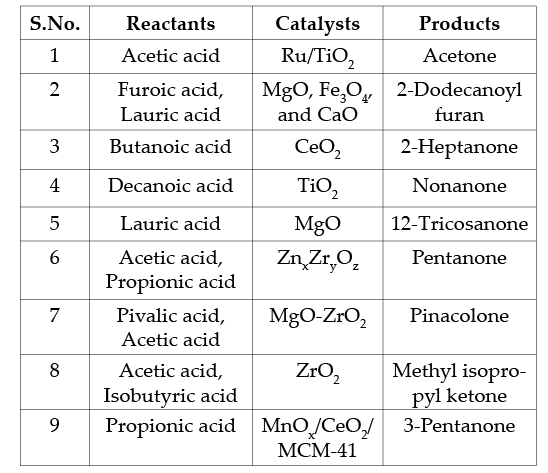
Ketonization of several carboxylic acids was reported over metal oxides, metals, zeolites, and bifunctional catalysts[7]. Metal oxide catalysts consist of highly basic oxides (MgO, CaO, BaO, etc.) with low lattice energy, and amphoteric oxides (TiO2, CeO2, ZrO2, etc.), having high lattice energy, are generally used as catalysts for ketonization reaction. Ketonization follows both bulk and surface mechanisms on solid catalysts based on the nature of the oxide. Surface ketonization is favorable over high lattice energy oxides, and the bulk mechanism occurs in basic metal oxides. Ketonization of different aliphatic and aromatic acids results in different ketones. Ketones such as diethyl ketone, acetophenone, propiophenone, pinacolone, 2-dodecanoyl furan, 12-tricosanone, etc, can be readily synthesized through the ketonization reaction. Different catalysts used to synthesize various ketones are summarized in Table 2. For example, the cross-ketonization of furoic acid and lauric acid in the presence of catalysts like MgO, Fe3O4, and CaO resulted in
2-dodecanoyl furan with a maximum yield of 90%. A vital chemical, pinacolone, was prepared by cross-ketonization of pivalic acid and acetic acid in a vapor phase fixed bed reactor using a mixed oxide catalyst of MgO-ZrO2: a combination of low-energy oxide and high-energy material[8]. The high content of ZrO2 in the catalyst was attributed to forming a mixed oxide MgZr1-xO2-x with increased basic sites, and showed the highest catalytic activity. In contrast, a comparative study of mixed oxide catalysts ZrO2, TiO2, and HfO2 for the ketonization of acetic acid showed that the formation of bidentate carboxylates on the ZrO2 surface reduced the activity as the reaction time increased[9].
Green Catalysis for Conversion of CO2 to Value-added Products
Reducing anthropogenic CO2 emissions and switching to renewable energy sources are the most important elements to mitigate climate change. Carbon Capture and Utilization (CCU) is a promising process for removing CO2 from industrial sources and converting it into value-added chemicals, including fuels, materials, and polymers. However, the relatively inert nature and low reactivity of CO2 present challenges for its conversion into valuable products. Therefore, the rational design of catalysts is essential for their activation and selective conversion. Synthesis of bulk and fine chemicals, such as formic acid and glycerol carbonate, through the catalytic conversion of CO2, is discussed in the section below.
CO2 to formic acid
Formic acid (FA) is one of the important CO2-derived products and has versatile applications in various industries like leather, cement, tanning, food, pharmaceuticals, etc[10]. It is also used as a potential medium for hydrogen storage due to its high volumetric capacity (53 g H2/L), low toxicity, and flammability under ambient conditions[11]. Commercially, formic acid is produced using toxic CO and methanol, with a worldwide production of ~800 kilotons per year. The current industrial production of FA has the potential to emit 3067 kg of CO2-eq per ton of FA production. If this technology is replaced by CO2-based production using renewable H2 sources, only 100 kg of CO2-eq per ton of FA would be expected to be emitted[12]. Thus, producing FA by CO2 hydrogenation is not only a sustainable route but also a greener process.
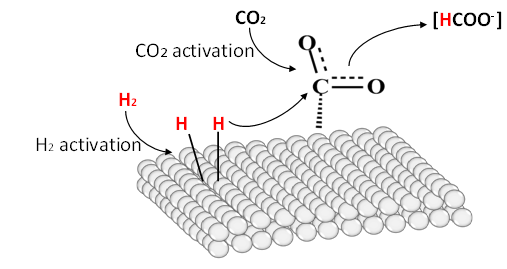
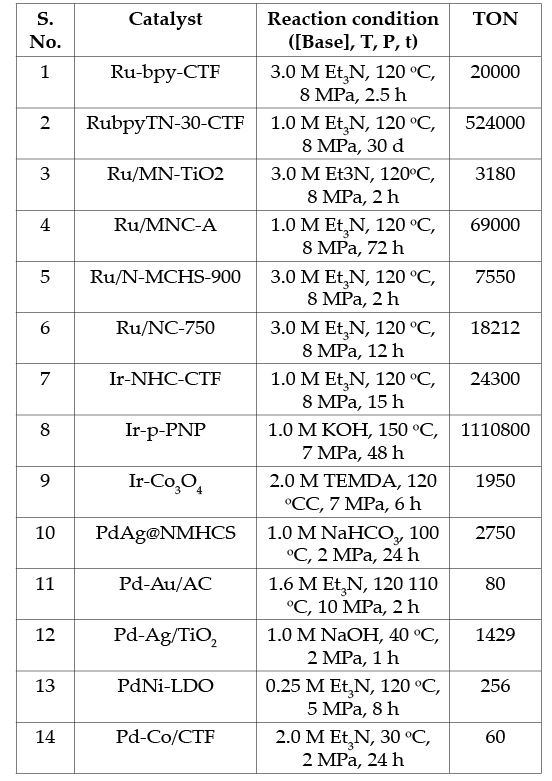
The hydrogenation of CO2 to formic acid is generally carried out in an aqueous solution with a base to overcome the thermodynamic limitations. Obviously, a catalyst must be needed to activate the relatively inert and low-reactive CO2and H2. The mechanistic pathway involves a multi-step mechanism where CO2 is adsorbed onto a metal catalyst, followed by hydrogen activation and dissociation to form metal hydride intermediates, which ultimately transfer the hydride to CO2 and desorb as a formate adduct, as shown in Scheme 2[13]. CO2 hydrogenation can be facilitated through both heterogeneous and homogeneous catalysis. However, heterogeneous catalysis offers distinct advantages, including enhanced catalyst stability, easier isolation, and reuse. In contrast, catalyst extraction and reusability challenges often hinder homogeneous catalysis. Post-reaction catalyst separation is essential to prevent potential reverse reactions during product separation, safeguarding process efficiency and product yield. Accordingly, numerous heterogeneous catalysts have been explored to mediate the hydrogenation in batch processes[14-16]. The catalytic performance of supported-metal catalysts strongly depends on the metal species’ local electronic structure and the support’s basicity. Many different types of heterogeneous catalysts, such as supported mono-metal catalysts, such as Co, Ni, Ru, Pd, Ir, and bimetal catalysts like PdAg, PdAu, PdNi, PdMn, PdCo, RuMn, RuCo, IrCo were explored (Table 3). Among others, heteroatoms-fused carbon supports with Ru showed excellent stability, with a maximum CO2 conversion of 45% and a turnover number (TON) of >524000 in relatively mild conditions (120 °C). Continuous hydrogenation of CO2 to formic acid in a fixed-bed reactor was also developed using heterogeneous Ru, Ir, and Pd catalysts. A pilot-scale study on converting CO2 to formic acid was recently reported at a 1 ton/day scale, with 82% CO2 conversion using a Ru-supported covalent triazine framework catalyst[17]. Further, the process delivers a 37% reduction in cost with a 42% decrease in global warming potential, demonstrating a feasible pathway toward commercial scalability and the advancement of the hydrogen economy.
CO2 utilization: Carbonylation of Glycerol to Glycerol Carbonate
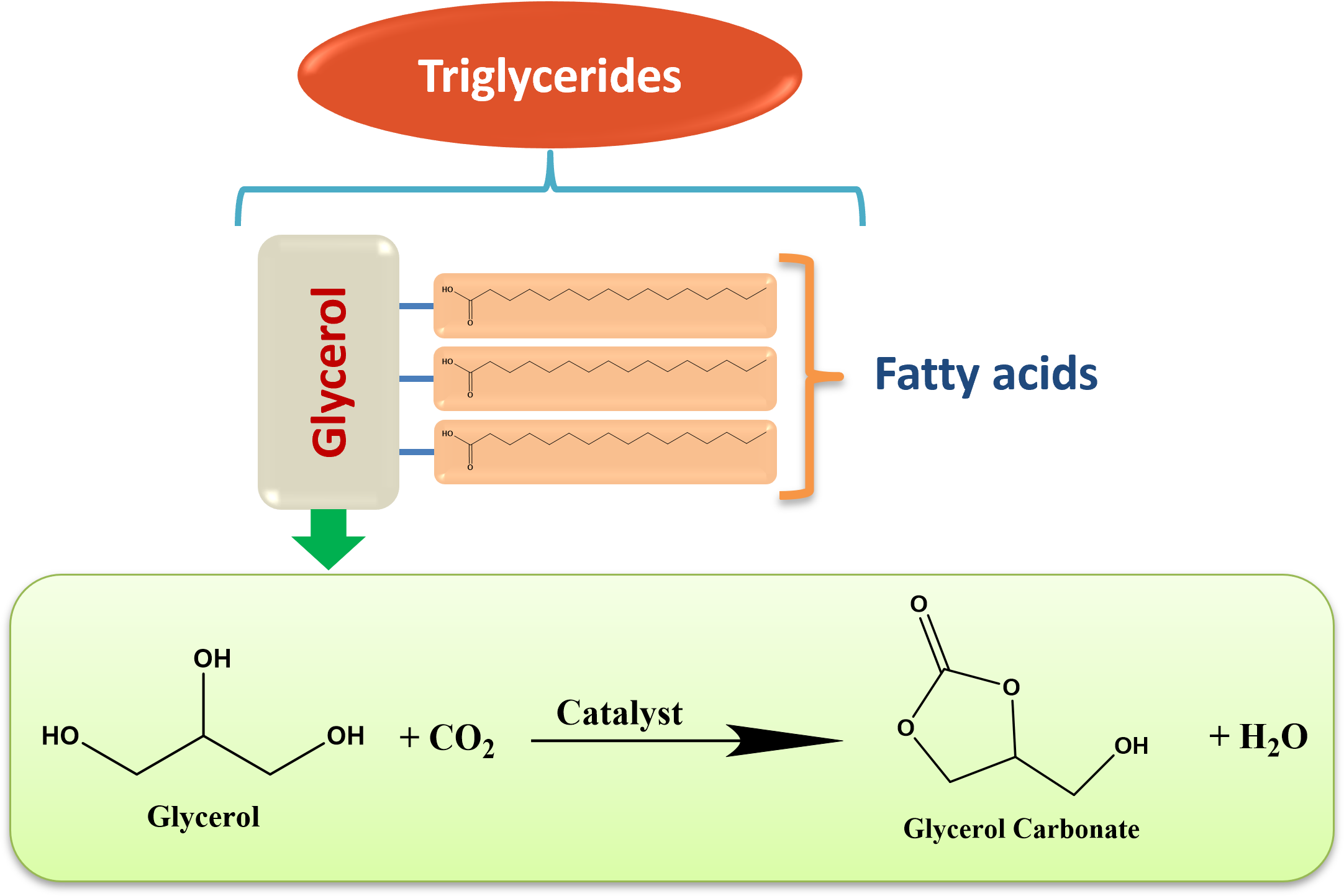
Glycerol is a by-product in biodiesel production, and it has a wide range of applications in various industries such as food, pharmaceutical, cosmetics, etc[18]. It is a cheap raw material for the production of fine chemicals by easy transformation reactions, including hydrogen production via steam reforming, hydrogenolysis to produce 1,2-propanediol, acetylation to produce monoacetin, etherification reactions to produce glycerol esters, carbonylation reaction to produce glycerol carbonate (GC), etc[19]. GC is an excellent raw material for applications due to its highly reactive hydroxyl and cyclic carbonate sites. GC has been used in different reactions as a green solvent, alkylating agent during the synthesis of β-Aryloxy alcohols, and several important intermediates of polymers and pharmaceuticals[18].GC can be synthesized by various routes, such as transesterification and carbonylation of glycerol by using ethers, phosgene-based methods, urea, and CO2 as carbonylation agents. Using CO2 as a carbonylation agent is more advanced, demonstrating high interest in Net Zero emissions, showcasing carbon neutrality, and helping to mitigate carbon pollutants from the atmosphere (Figure 3).
The catalytic carbonylation of glycerol using CO₂ is a green chemistry approach, and it has several challenges in developing a catalyst due to the high thermodynamic stability of CO₂. Understanding the general mechanism of glycerol to GC is required to develop a highly stable and active catalyst. Generally, the glycerol is adsorbed on the Lewis basic sites of the catalyst. CO2 is activated on the oxygen vacancies or the mobile oxygen in the form of carbonate intermediates. The adsorbed glycerol and the activated CO2 form a seven-membered ring of ester intermediate. In the final step, an intramolecular nucleophilic attack occurs where the alkoxy group reacts with the carbonyl carbon atoms to produce GC. Understanding the mechanism can help deduce the active sites required for the catalyst design. As the reaction requires Lewis basic sites and the mobile oxygen to activate the two reactants, the basic oxide materials are studied extensively. On the other hand, the availability of mobile oxygen can be easily achieved by creating defects in metal oxides such as CeO2[20]. The catalytic conversion of glycerol to GC with CO2 is studied on different mono-metallic and bi-metallic catalysts, in which prominent catalysts are CuO, MgO, CaO, Al2O3, CeO2, MgO-CaO, MgO-Al2O3, CaO-Al2O3, and CeO2-ZrO2. Among all the mono-metallic catalysts, CuO showed a high rate of product formation[21,22]. On the other hand, Zn-modified CeO2 catalysts showed an activity of 90% of both conversion and yield of glycerol and GC[23]. In our recent work, we developed a ternary oxide catalyst designed using MgO-ZnO-CeO2 oxides that showed a promising activity of 90% conversion of glycerol, and the formation rate of product GC is 4.6 mmolg-1h-1 [24].
Summary
The field of catalysis has shown a high impact in achieving sustainability goals and green chemistry processes with their unique applications in various reactions, like synthesizing value-added chemicals and fuels from biomass and CO2. The catalytic conversion of biomass to sustainable aviation fuel is an eco-friendly process for producing alternative fuels. The catalytic ketonization of acids is a green and sustainable process compared to the traditional acylation reactions using toxic chloride-based materials. The synthesis of fine and bulk chemicals, fuels, and fuel additives can be achieved by CO2 utilization reactions, in which the design and development of green catalytic materials play a vital role in the activation of CO2. Designing catalysts with the desired properties for specific reactions is essential for achieving green catalytic processes.
References
[1] S. Mailaram, P. Kumar, A. Kunamalla, P. Saklecha, S.K. Maity, in: Sustain. Fuel Technol. Handb., INC, 2021, pp. 51–87.
[2] W. Wang, N. Li, S. Li, G. Li, F. Chen, X. Sheng, A. Wang, X. Wang, Y. Cong, T. Zhang, Green Chem. 18 (2016) 1218–1223.
[3] A. Kunamalla, S. Mailaram, B.S. Shrirame, P. Kumar, Hydrocarbon Biorefinery: A Sustainable Approach, Elsevier Inc., 2022.
[4] A. Kunamalla, S.K. Maity, Fuel 332 (2023).
[5] G. Li, N. Li, Z. Wang, C. Li, A. Wang, X. Wang, ChemSusChem 5 (2012) 1958–1966.
[6] T. Goculdas, S. Deshpande, W. Zheng, R.J. Gorte, S. Sadula, D.G. Vlachos, Green Chem. 25 (2022) 614–626.
[7] B. Boekaerts, B.F. Sels, Appl. Catal. B Environ. 283 (2021) 119607.
[8] P.M. Kumar, Y. Nagesh, G. Mallikarjun, N. Lingaiah, Catal. Letters (2024).
[9] P. Tingelstad, R.L. Peters, S.K. Regli, A. Miró I Rovira, K. Rajendran, D. Chen, Catal. Today 443 (2025) 114961.
[10] W. Reutemann, and H.Kieczka, Formic Acid. In Ullmann’s Encyclopedia of Industrial Chemistry; Wile y-VCH: Weinheim, Germany, 2011.
[11] M. Grasemann, and G.Laurenczy, Energy Environ. Sci. 5 (2012) 8171−8181
[12] N. von der Assen, P. Voll, M. Peters and A. Bardow, Chem. Soc. Rev. 43 (2014) 7982–7994
[13] G. H. Gunasekar, K. Park, K.-D. Jung and S. Yoon, Inorg. Chem. Front., 2016, 3, 882-895
[14] G. H. Gunasekar, J. Shin, K.-D. Jung, K. Park and S. Yoon, ACS Catalysis 8 (2018) 4346-4353
[15] K. Park, G. H. Gunasekar, S.-H. Kim, H. Park, S. Kim,K. Park, K.-D. Jung and S. Yoon, Green Chem, 2020, 22, 1639-1649
[16] Z. Wang, D. Ren, Y. He, M. Hong, Y. Bai, A. Jia, X. Liu,C.Tang, P. Gong, X.Liu, W. Huang, and Z. Zhang ACS Catal. 2023, 13, 10056−10064
[17] C. Kim, K. Park, H. Lee, J. Im, D. Usosky, K. Tak, D. Park, W. Chung, D. Han, J. Yoon, H. Lee, H. Kim, Margareth, J. Jung, D. Hye Won, C.-J. Yoo, Ki Bong Lee, K.-D. Jung, and U. Lee, Joule 8, (2024) 1–21.
[18] P. Inrirai, J. Keogh, A. Centeno-Pedrazo, N. Artioli, H. Manyar. J. CO2 Util 80 (2024) 102693.
[19] S. Sahani, S.N. Upadhyay, Y.C. Sharma.Int. J. Hydrogen Energy, 41 (2016) 2285–2297.
[20]F. Strniša, V. T. Sagar, P. Djinović, A. Pintar, I.Plazl. Chem. Engg. J, 413 (2021) 127498.
[21] Y. Ke, H. Xu,X. Wang, H. Liu, H. Yuan.J. CO2 Util.83(2024) 102813.
[22] J. Liu, Y. Li, J. Zhang, D. He. Appl. Catal. Gen. 513 (2016) 9-18.
[23] Kulal, N.; Vetrivel, R.; Ganesh Krishna, N. S.; Shanbhag, G. V. ACS Appl. Nano Mater. 4 (2021) 4388-4397.
[24] P. Pattanaik, M. Geekuri, K. Ranjithkumar, G. H. Gunasekar, L. Nakka. ACS Appl. Nano Mater. 7 (2024) 3580-23591.
 Dr. N.Lingaiah (PhD) is Chief Scientist and chair of the Department of Catalysis and Fine Chemicals, CSIR-Indian Institute of Chemical Technology, Hyderabad. He has worked as a postdoctoral fellow in South Korea, Japan, and the UK. He is also a visiting scientist at Lehigh University, Bethlehem, United States, and IRCELYON, France. He has over 30 years of experience in the field of heterogeneous catalysis. He was awarded the BOYSCAST and Raman Research fellowship and is also a Fellow of Telangana Academy of Sciences.
Dr. N.Lingaiah (PhD) is Chief Scientist and chair of the Department of Catalysis and Fine Chemicals, CSIR-Indian Institute of Chemical Technology, Hyderabad. He has worked as a postdoctoral fellow in South Korea, Japan, and the UK. He is also a visiting scientist at Lehigh University, Bethlehem, United States, and IRCELYON, France. He has over 30 years of experience in the field of heterogeneous catalysis. He was awarded the BOYSCAST and Raman Research fellowship and is also a Fellow of Telangana Academy of Sciences.

Dr. K. Alekhya (PhD) is a scientist in the Department of Catalysis and Fine Chemicals, CSIR-Indian Institute of Chemical Technology, Hyderabad. She specializes in using heterogeneous catalysis in biorefineries to produce fuels and value-added chemicals.
 Dr. G.H. Gunasekar (PhD) is a scientist in the Department of Catalysis & Fine Chemicals, CSIR-Indian Institute of Chemical Technology, Hyderabad. He worked as a senior research officer at the IOCL R&D Centre, Faridabad, and at Orchid Chemicals and Pharmaceuticals R&D Centre. His area of research interest includes the valorization of CO2 and biomass into value-added chemicals and fuels like formic acid, methanol, etc., as well as the development of green and sustainable processes for various catalytic conversions.
Dr. G.H. Gunasekar (PhD) is a scientist in the Department of Catalysis & Fine Chemicals, CSIR-Indian Institute of Chemical Technology, Hyderabad. He worked as a senior research officer at the IOCL R&D Centre, Faridabad, and at Orchid Chemicals and Pharmaceuticals R&D Centre. His area of research interest includes the valorization of CO2 and biomass into value-added chemicals and fuels like formic acid, methanol, etc., as well as the development of green and sustainable processes for various catalytic conversions.
 Dr. Tatiparthi Vikram Sagar (PhD) is a scientist in the Catalysis and Fine Chemicals Department at CSIR-Indian Institute of Chemical Technology in Hyderabad. He has worked as a postdoctoral research fellow in Slovenia at the University of Ljubljana and the National Institute of Chemistry, Slovenia. His research is focused on CO2 utilization reactions for producing hydrogen and bulk chemicals, such as methanol, formic acid, and acetic acid.
Dr. Tatiparthi Vikram Sagar (PhD) is a scientist in the Catalysis and Fine Chemicals Department at CSIR-Indian Institute of Chemical Technology in Hyderabad. He has worked as a postdoctoral research fellow in Slovenia at the University of Ljubljana and the National Institute of Chemistry, Slovenia. His research is focused on CO2 utilization reactions for producing hydrogen and bulk chemicals, such as methanol, formic acid, and acetic acid.

































